Tuning the Brain: How Electricity, Magnetism, and Sound Waves Reshape Neural Circuits
Thursday, 12 June 2025
In our previous post in this series, we explored how the brain's neural circuits operate—how neurons communicate through electrical impulses, maintain the critical balance between excitation and inhibition, and form the networks that underlie all brain function. But understanding how the brain works naturally is only the first step. The next question becomes: how can we intentionally influence these circuits when they malfunction?
This challenge has driven decades of innovation in neuromodulation technologies. Today, we have multiple ways to deliver energy to the brain—electrical, magnetic, and acoustic—each interacting with neural tissue through distinct physical mechanisms. Understanding these differences is crucial for appreciating why certain approaches work better for specific conditions and why the field continues to evolve toward more precise targeting methods.
The Physics of Influencing Neurons
Before diving into specific technologies, it's worth understanding what all brain stimulation methods have in common: they work by altering the electrical properties of neurons. Every neuron maintains a voltage difference across its membrane—the "resting potential"—and brain stimulation techniques essentially manipulate this electrical environment.
Think of a neuron like a tiny battery that's constantly being charged and discharged. The cell maintains this electrical state through ion channels—specialized proteins that control the flow of charged particles (ions) like sodium, potassium, and calcium across the cell membrane. When enough positive charge enters the neuron to reach a critical threshold, it fires an action potential. Sodium rushing in triggers the neuron to fire, while potassium flowing out helps reset it—like a biological on/off switch.
We can influence this process with external energy sources in two fundamental ways: we can either nudge neurons closer to or further from their firing threshold without actually triggering an action potential (subthreshold effects), or we can deliver enough energy to directly cause neurons to fire (suprathreshold effects). This distinction is crucial for understanding how different technologies work and why they're suited for different applications.
Electrical Stimulation
When we apply electrical stimulation to the brain, we're taking the most direct approach to influencing neural circuits—using electrical current to change the voltage environment around neurons. However, the three main electrical approaches differ dramatically in their precision, invasiveness, and mechanisms of action.
Transcranial Direct Current Stimulation (tDCS): Broad Network Modulation
With tDCS, we apply a weak electrical current (typically 1-2 milliamps) through sponge or gel electrodes placed on the scalp. These electrodes—classically, each about the size of a playing card and soaked in saline solution—connect to a battery-powered device roughly the size of a smartphone. This might seem like a lot of current, but by the time it passes through the skull and reaches the brain, the actual electric field strength is quite modest—shifting neurons' voltage by just 0.1 to 0.5 millivolts, much less than what is required to trigger an action potential.
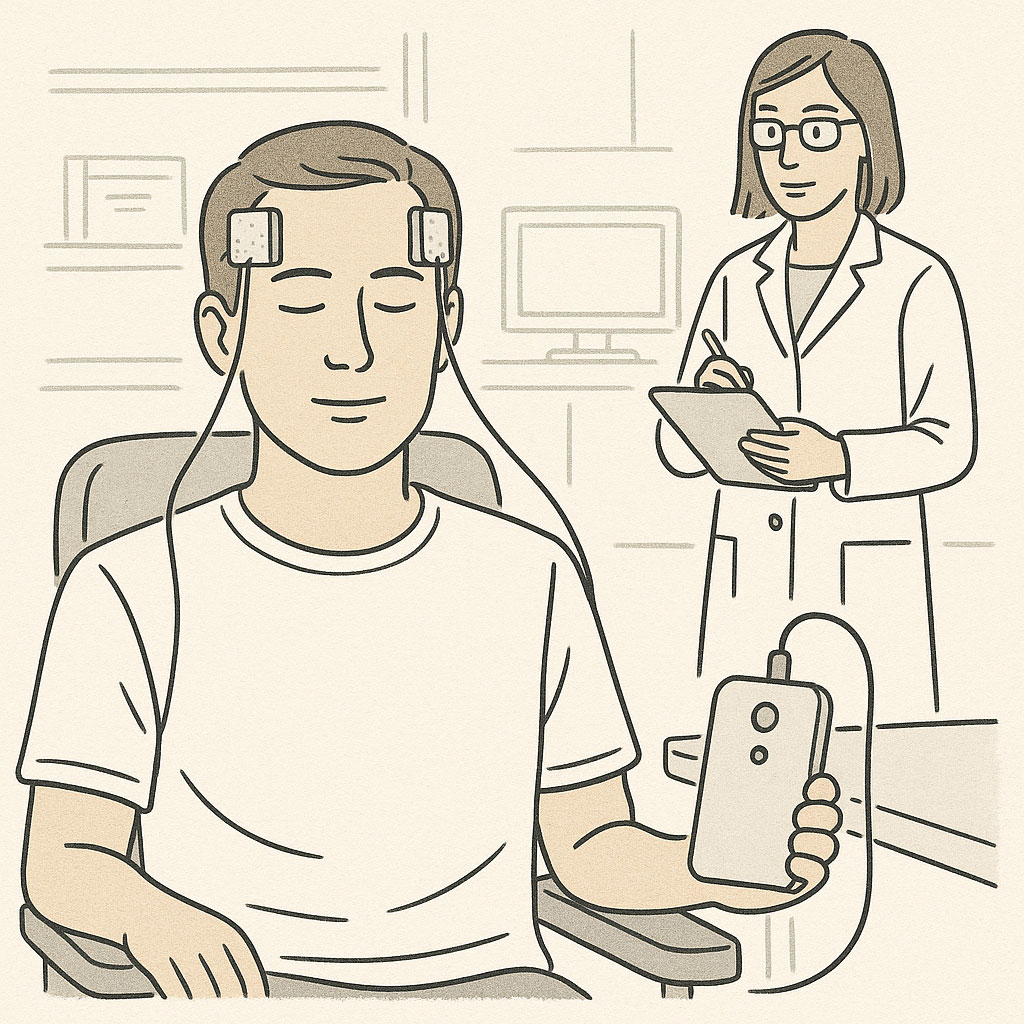
Transcranial direct current stimulation (tDCS) uses electrodes on the scalp to deliver weak electrical current, gently shifting neural excitability across broad brain regions.
The key insight about tDCS is that it works through subthreshold modulation—gently biasing neural activity instead of commanding it. We don't force neurons to fire; we shift their resting potential, making them either more likely or less likely to fire when they receive normal synaptic inputs. Imagine adjusting the sensitivity on a smoke detector—you're not setting off the alarm directly, but you're changing how easily it will trigger when it encounters smoke.
Since the electrical current spreads diffusely through brain tissue, tDCS influences broad cortical areas, though the strength and consistency of its effects are modest and vary across individuals. This broad influence has made it a candidate for conditions where we want to gently shift the excitability of entire networks.
Deep Brain Stimulation (DBS): Surgical Precision
At the other end of the spectrum, deep brain stimulation requires a sophisticated surgical system. The visible components include a thin wire electrode (about 1.3 mm in diameter) implanted directly into specific brain targets, connected via an extension cable tunneled under the skin to a pulse generator—similar to a cardiac pacemaker—typically implanted below the collarbone.

Deep brain stimulation (DBS) delivers precise electrical pulses through a surgically implanted electrode connected to a pulse generator in the chest.
Once activated, this system delivers continuous high-frequency electrical pulses (typically 130-185 Hz) around the clock, with settings adjustable via an external programmer. The remarkable precision of DBS comes from placing these electrodes just millimeters from target neurons, allowing focused stimulation of specific neural populations—like targeting a specific floor in a skyscraper rather than the whole building.
In conditions like Parkinson's disease, this surgical precision proves invaluable. We can target specific nuclei in the brain's motor control circuits that have become dysfunctional, with the high-frequency electrical pulses disrupting pathological synchrony—when neurons fire together in harmful lockstep patterns, like a crowd chanting in unison when they should be having individual conversations.
Cortical Electrode Arrays: Cellular-Level Interface
For the highest level of electrical precision, we turn to cortical electrode arrays—grids of tiny electrodes placed directly on or within brain tissue. These systems allow us to interact with extremely small groups of neurons, sometimes down to the level of individual cells.
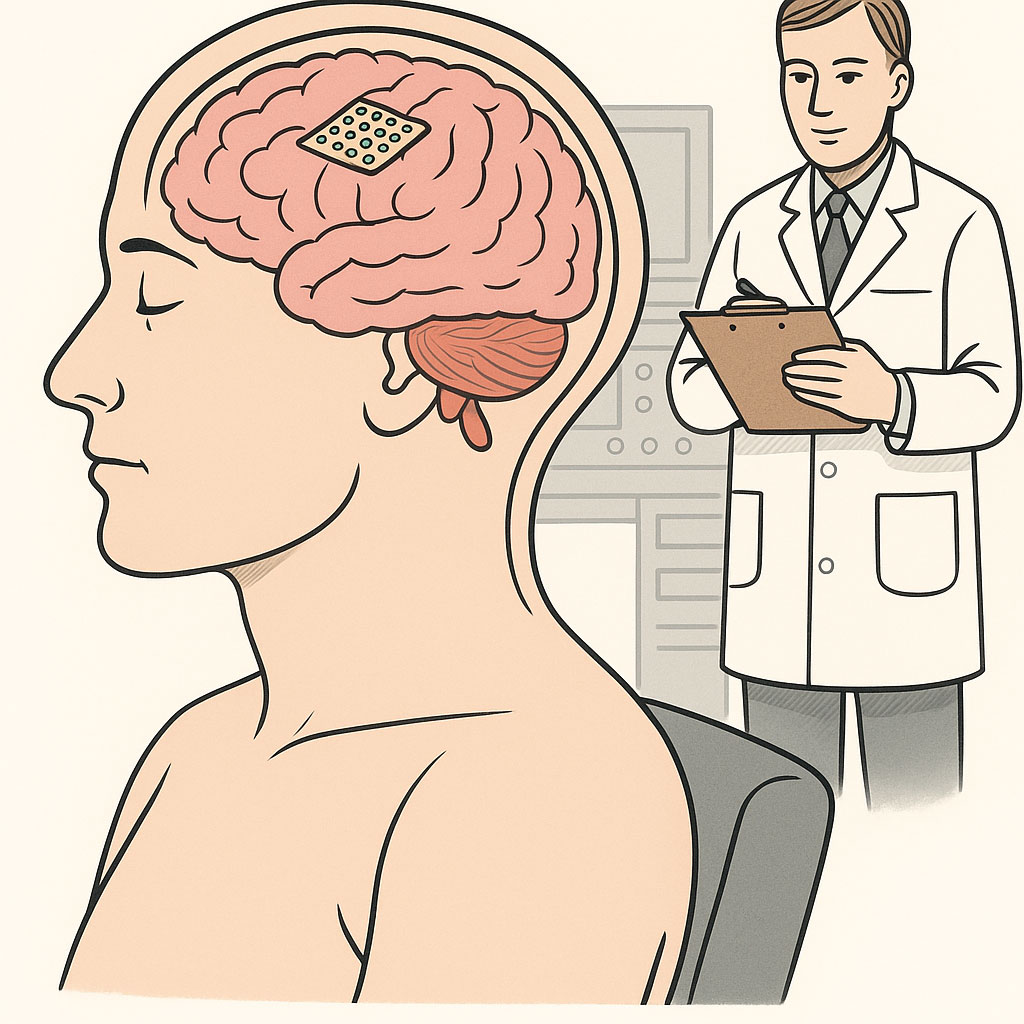
Electrode arrays placed directly on brain tissue enable precise monitoring and stimulation of small groups of neurons.
Electrode arrays take various forms: flexible grids that conform to the brain's surface, rigid microelectrode arrays with needle-like projections, or thin strips that can slide into deep brain folds. A typical grid might contain 64 to 256 individual electrode contacts, ranging from sub-millimeter to a few millimeters, all embedded in biocompatible material.
The direct tissue contact means very low currents can be used while maintaining exceptional precision. Each electrode might influence only hundreds of neurons, enabling researchers and clinicians to test specific circuit functions with unprecedented resolution.
Magnetic Stimulation: Induction at a Distance
Moving from direct electrical approaches, we encounter a fundamentally different strategy with transcranial magnetic stimulation (TMS). Rather than applying electrical current directly, TMS uses rapidly changing magnetic fields to induce electrical currents within brain tissue—a phenomenon described by Faraday's law of electromagnetic induction.
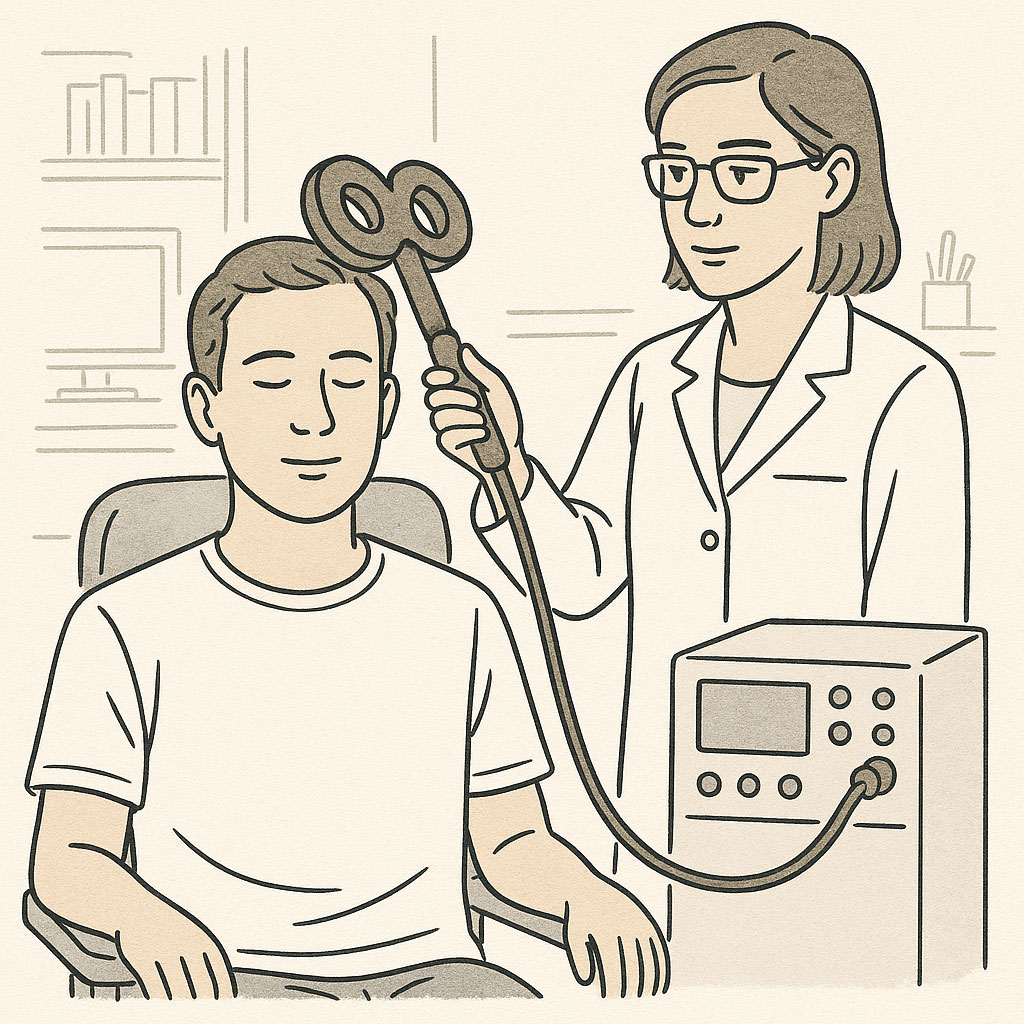
Transcranial magnetic stimulation (TMS) uses a figure-8 shaped magnetic coil positioned against the scalp to induce electrical currents in targeted brain regions.
During TMS treatment, we position a magnetic coil—typically shaped like a figure-8 and about the size of a small dinner plate—against specific areas of the scalp. This coil generates a powerful, brief magnetic pulse that passes harmlessly through the skull and induces circular electrical currents in the underlying brain tissue. With each pulse, the device produces a distinctive clicking sound. We can tune these induced currents to either trigger action potentials (suprathreshold) or subtly modulate neural excitability (subthreshold), with most therapeutic protocols using suprathreshold intensities.
The physics of magnetic induction gives TMS some unique characteristics. While the magnetic field passes through the skull relatively unimpeded, the strength of the induced electrical currents drops off rapidly with distance from the coil. This physical constraint means TMS primarily affects superficial cortical areas—typically the outer 2-3 centimeters of the brain—like illuminating the top floors of our neural skyscraper while leaving the basement relatively untouched.
Despite this depth limitation, TMS has found particular success in treating depression, where stimulation of specific cortical areas can cascade down to influence deeper brain networks involved in mood regulation. The ability to deliver precise, timed pulses has also made TMS valuable for mapping brain function, allowing scientists to temporarily disrupt specific brain areas and observe the effects.
Ultrasound Stimulation: Mechanical Modulation
The newest player in the brain stimulation field takes yet another approach, using focused acoustic energy—sound waves at frequencies far above human hearing. When we use transcranial ultrasound stimulation, we're employing a fundamentally different mechanism compared to electrical or magnetic approaches.
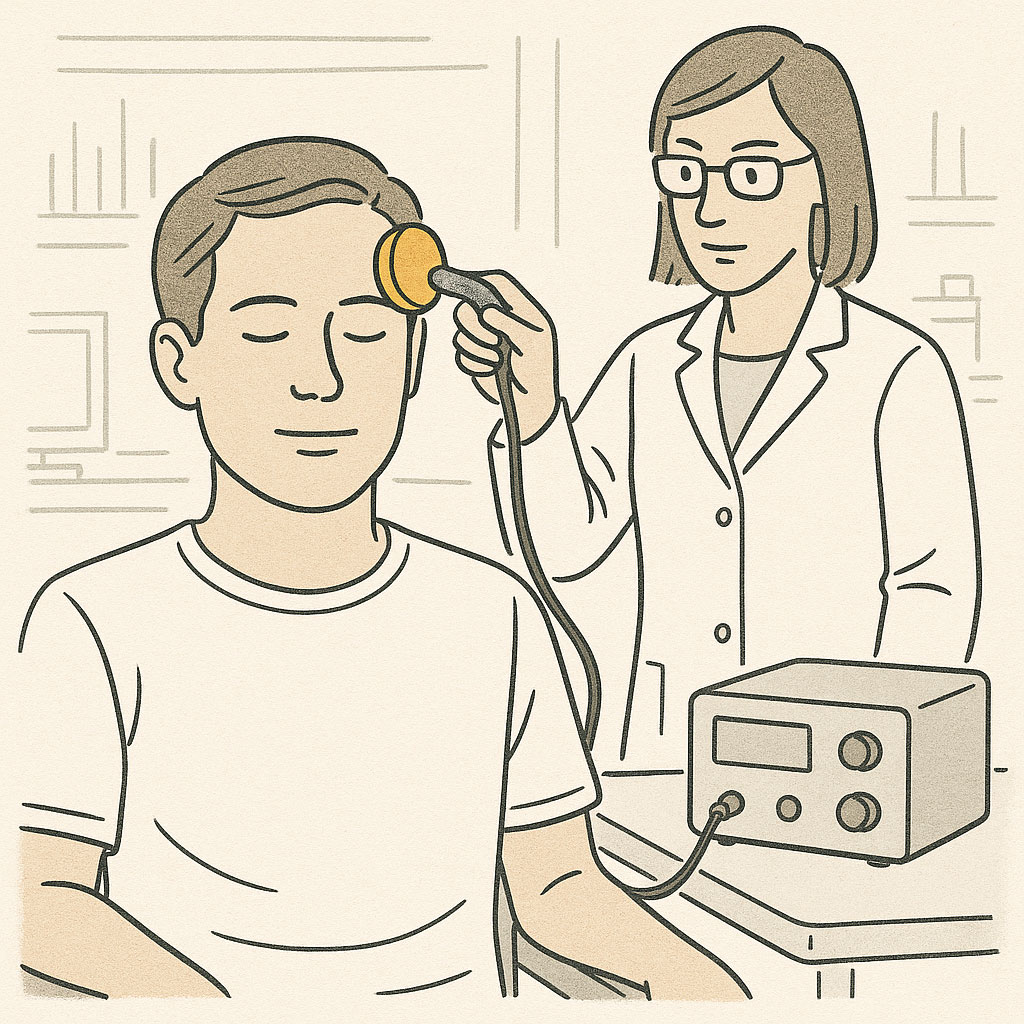
Focused ultrasound uses high-frequency sound waves to mechanically influence neural activity with millimeter precision, even in deep brain structures.
Instead of directly manipulating electrical fields, ultrasound creates mechanical pressure waves. These sound waves produce tiny mechanical deformations in neural tissue—imagine gently tapping on neurons many thousands of times per second. This mechanical stimulation is thought to influence specialized proteins called mechanosensitive ion channels, which respond to physical forces by opening or closing their gates, ultimately affecting the neuron's electrical state—though the exact cellular and circuit-level mechanisms remain under investigation.
This mechanical approach to neural modulation—using physical pressure rather than electrical fields—offers some advantages. We can focus sound waves much more precisely than electrical or magnetic fields, potentially achieving spatial resolution comparable to invasive methods while remaining completely non-invasive.
Additionally, ultrasound possesses a unique capability among non-invasive methods: it can reach deep brain structures that other approaches struggle to access. The ability to focus acoustic energy to volumes as small as a few millimeters while penetrating deep into the brain represents a significant advance, though we're still working to fully understand and validate these capabilities in clinical settings.
Comparing the Approaches
Each stimulation modality reflects different trade-offs between precision, accessibility, and mechanism of action. As we've seen, these technologies vary dramatically—from tDCS's gentle, widespread modulation to electrode arrays' precise cellular control, from TMS's magnetic induction to ultrasound's mechanical waves. Understanding these differences helps explain why certain approaches suit specific clinical needs:
| Method | Energy Type | Precision | Invasiveness | Primary Mechanism |
|---|---|---|---|---|
| TDCS | Electrical | Low | Non-invasive | Subthreshold |
| TMS | Magnetic | Moderate | Non-invasive | Suprathreshold |
| DBS | Electrical | High | Invasive | Suprathreshold |
| Ultrasound | Acoustic | High | Non-invasive | Subthreshold (via mechanical) |
| Electrode Arrays | Electrical | Very High | Invasive | Suprathreshold |
This comparison reveals the central challenge we face in neuromodulation: historically, achieving high precision has required invasive procedures, while non-invasive methods have been limited in their targeting capabilities. Technologies like focused ultrasound represent promising efforts to break this trade-off, potentially offering the precision of surgical approaches through completely non-invasive means.
Looking Ahead
Understanding these different mechanisms reveals the central challenge facing brain stimulation: matching the right tool to the right problem. For some disorders, broad network modulation might be exactly what's needed. For others, millimeter precision could make the difference between success and failure.
But precision is only one dimension. The timing and patterns of stimulation matter too—whether we deliver continuous pulses or bursts that mimic natural brain rhythms can determine whether effects last minutes or months. How different approaches promote lasting brain changes through neuroplasticity is a fascinating topic for another post.
In upcoming posts, we'll dive deep into this question of spatial precision—examining why the field is pushing toward increasingly targeted approaches and how this shapes which conditions each technology can effectively treat.
The development of these diverse neuromodulation technologies represents one of the most exciting frontiers in neuroscience. By understanding how different forms of energy interact with neural circuits, we gain insights not only into how the brain works, but also into how we might restore function when these remarkable circuits malfunction.
This post is the second in a series exploring brain stimulation technologies. Future posts will examine how the spatial precision of different approaches determines their clinical applications and effectiveness.